Biology 441, Spring 2013: Lecture Notes on Cell Forces
Physical Forces: (Please read Chapter Eight of our textbook)
What forces create anatomical shapes and patterns?
→ NOT growth pressure, somewhat surprisingly!
(I will come back to this issue.)
→ Acto-myosin contraction. (Very important; and very diverse)
-
Examples:
-
Bending of epithelia by apical contraction of acto-myosin.
-
"Apical Constriction"
Invagination, Involution, Ingression.
(mitotic cytokinesis)
Locomotion of cells and epithelial cell sheets.
"Cell Motility", or "Crawling"
By exertion of traction tangentially through plasma membranes.
Retrograde Transport
...of anything attached to outside of the plasma membrane.
Actin fibers continually assemble from diffusing actin monomers.
Also inflating the eyeball, and the cornea.
-
Pressure = Tension x Curvature as I will explain soon
The lower the tension, the more surfaces bulge out.
(And this is a linear proportionality, too!)
(But don't worry about the math; the ideas will be sufficient.)
→ Electro-osmotic pressure. especially in Cartilage.
(This will help you revolutionize Orthopedic Surgery!)
How does cytoplasmic actin exert traction forces by which tissue cells crawl (including tissue culture cells) ?
Most cell biology textbooks describe the "amoeboid" locomotion of body cells as if it were produced by protrusion of filopodia, attachment filopodia to some external object, and then contraction of the filopodia. This is supposed to pull the cell forward. It is a fiction; it doesn't happen. Just look at a time-lapse video of tissue culture cells and it is obvious that is not how they propel themselves. Unfortunately, the NIH has spent many millions of dollars for a large research program based on this fiction. It concentrates on identifying cytoplasmic proteins that become concentrated near the leading margins of fibroblasts and other tissue cells. These proteins are sure to be important, but it is impossible that they serve for force exertion by forward protrusion or rearward retraction of filopodia, since that method of cell propulsion is imaginary. Much direct evidence disproves that protrusion-retraction theory of cell motility. For example, retrograde transport of attached particles disproves that theory. Another disproof is the location where traction is exerted by cells crawling on flexible gels or thin sheets of rubber.
The actual mechanism of cell crawling if that cytoplasmic actin monomers assemble into fibers along the leading (front, but also rear) edges of a cell. These actin fibers flow "rearward" toward the center of the cell (nearer to the nucleus). Think of a tractor tread that is inside a large
sack, and has to exert sideways forces through the material of this sack, from inside to outside. This sideways transmission of force is one of many phenomena that were made understandable by the Singer-Nicholson fluid-mosaic explanation of membrane structure. A chain of connected proteins, of which fibronectin and integrins are most important, transmit physical connections from the rearward-flowing actin fibers inside the cell to adhesions outside the cell.
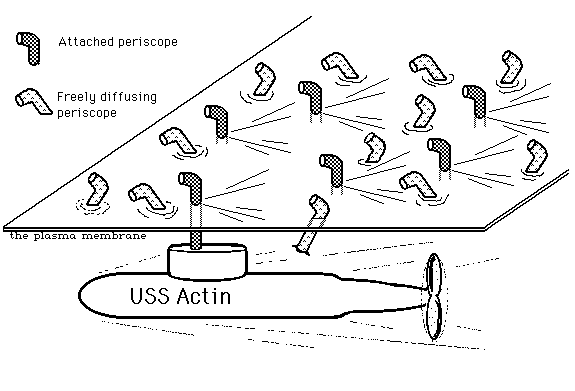
Visualize a fleet of many submarines, all traveling along together in the same direction, with their periscopes sticking up through the surface. The submarines represent the moving actin fibers; Their periscopes represent the trans-membrane exertion of forces by integrins and fibronectin. Finally, visualize ice-bergs being pushed directionally across the ocean surface by sideways pressure of the the periscopes. You can hardly blame people for preferring to imagine that body cells crawl by a cycle of protrusion, attachment, and contraction of filopodia and other outward extensions of advancing cell margins. So what if that is inconsistent with where crawling cells exert forces, and contradicted by rearward transport of particles or any other movable object attached to the outside of the plasma membrane.
-----------------------------------------------------------------------------
Please read the following excerpt from a review paper by Dr. Louise Cramer, a British Cell Biologist: (The entire paper is available on line, and conveys a (still-up-to-date!) good impression of the difficulties and debates about this subject. It will show you the need and opportunity for more research and careful thinking on this subject.)
[Frontiers in Bioscience, 2, d260-270, June 1, 1997]
MOLECULAR MECHANISM OF ACTIN-DEPENDENT RETROGRADE FLOW
IN LAMELLIPODIA OF MOTILE CELLS
Louise P. Cramer
The Randall Institute, Kings College London, 26-29 Drury Lane, London WC2B 5RL, UK.
The following is a short quote from near the middle of Dr. Cramer's paper:
In this review, I will briefly describe the organization of actin filaments in leading edge structures of adherent, motilearrow to left) and lamella (dashed thick arrow to left), and recently, mostly cells, and in neuronal growth cones. Then, I will describe potential types of actin-dependent motile force to drive retrograde particle flow relative to the substratum in lamellipodia of these cells, and in growth cones of Aplysia neurons. I will present evidence in favor of each type of motile force, and discuss function of retrograde actin flow"...etc.
-----------------------------------------------------------------------------
In conclusion, let's now briefly return, to the submarine & periscope analogy, regarding how actin fibers exert propulsive traction forces through the plasma membrane, from cytoplasm to extracellular objects. Several kinds of evidence indicate that the mechanical connection varies, somewhat as if the periscopes in the analogy attached and detached from their submarines.
Although difficult to visualize, such variations should have been expected. After all, binding
between all sorts of molecules is only temporary, with an equilibrium between bound and unbound molecules. Can you think of an example of molecular binding where that isn't true?
Please just give some thought to methods be which one could measure rates of turn-over in
actin-external binding, through the plasma membrane. Wolpert himself has had some very clever ideas on this subject.


"...Early ideas on how actin filaments generated force to drive retrograde particle flow in lamellipodia were theoretical. One quite popular idea was that contraction of an actin filament network moved the lipid bilayer of a lamellipodium backward as a sheet, and structures on the moving sheet rode as passengers (4, 11). At the time this made sense; flow of particles on the surface of lamellipodia were thought to reflect a moving cell surface, and muscle proteins were just beginning to be identified in non-muscle motile cells (reviewed in (12)). This theory was not pursued once Singer and Nicholson (13) introduced the idea that the lipid bilayer was fluid. Of the several alternative explanations offered, the one that turned out to be the most pertinent came from a discussion between Wolpert and Harris in 1973 (11). Wolpert hypothesized that a 'filamentous system' directly moved particles retrograde. Precisely how has been debated since this time. Part of the problem is that over the last 10 years or so different types of particles have been studied in different motile cell types. For example, the tendency has been to view particles flowing retrograde on the cell surface, as the same phenomenon as particles and actin filaments flowing retrograde inside the lamellipodium. It may turn out, however, that retrograde flow of particular types of particles associated with lamellipodia in some motile cell types, may be a separate phenomenon, driven by a distinct mechanism. Perhaps related to this, different results have been obtained in different motile cell types, particularly in Aplysia bag cell neuronal growth cones, fibroblasts and keratocytes. This has led to distinct views on both the mechanism of retrograde particle flow, and function of retrograde actin flow in lamellipodia.
I added the boldface and the underlining, for emphasis. They are not in the original paper.