Embryology Biology 441 Spring 2014 Albert Harris
Limb bud development

Photo: Cartilages differentiating inside a living chicken hind-limb bud
Limb buds develop into legs, arms, wings, or fins.
Normally, embryos form 4 limb buds,
2 anterior & 2 posterior.
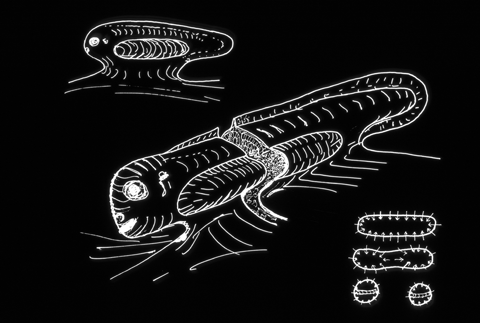
But are there ways to induce salamander embryos to form three legs on each side?
(Like the Martians in "John Carter of Mars"!)
Surgically implanting otic or olfactory placodes, or plastic beads containing Fibroblast Growth Factor, will cause three legs to form on the same side.
Scientists suspect that embryos re-use the same inductive signals in different parts of the body; and inductively competent is already concentrated along both flanks.
Limb buds develop if transplanted to other parts of the body, and to the amnion of chicken embryos.
You can also split limb buds, fuse limb buds, rotate them upside down, or backwards.
If you cut an early limb bud in two, it can form 2 arms, or often it will form a branched leg (mirror images).
Fusing two limb buds can often form only one leg. (surgically graft one limb bud right next to another one, & they merge! These kinds of experiments work best in salamander embryos.)
Notice the similarity to what Driesch discovered! That splitting early echinoderm embryos in two results in each half developing into a half-sized "scale model" embryo.
Several body organs have been discovered to form two organs, for example two complete hearts.
Another example is eyes, and also the nose rudiment, which can be either split or into 2, or two can be fused into one.
The human uterus also forms by side to side fusion of the lower end of the oviduct. (Muellerian Duct).
[Be prepared for an exam question: Describe at least ? embryonic organs that develop by fusion of two masses of cells. List examples of organs or organisms being split into two, where each half is able to form all parts normally developed when they are not split.]
Some kinds of parasitic worms often split limb buds in frogs, resulting in 5 or 6 hind limbs!
There is a color photograph of this in every edition of Scott Gilbert's textbook.
&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&
Development of the 3 geometric axes are controlled separately in limb buds And at 3 different times, by 3 different sets of chemical signals
-
Proximo-distal axis
Anterior-posterior axis
Dorso-ventral axis
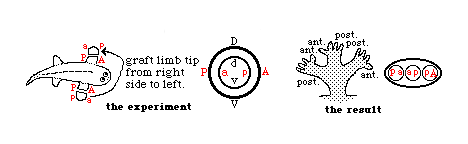
During a later period of development, limb buds are no longer able to regulate (change) their anterior-posterior axis, but can still regulate the dorso-ventral axis!
In effect, that means that the part that would have developed into the palm of the hand can change its fate, and develop the geometry appropriate to the back of the hand, and vice versa, but a rotated limb bud can no longer change where the thumb forms, etc.
Analogous surgical experiments were done with the inner ear (otic placode) cutting it out and grafting it back in, upside down and backwards, etc.
Before a certain stage, it "regulates" completely, in the sense that the different cells change how they develop according to their new orientations, such that the semi-circular canals, otoliths, etc. all develop at locations that are normal with respect to the rest of the body.
At a later stage of development, the dorso-ventral and medio-lateral axes are still able to regulate, but the anterior-posterior axis is irreversible.
These rotation experiments were done in salamanders, in the 1910s-30s.
(the limb bud experiments and also the inner ear experiments, and many others.)
&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&&
The Apical Ectodermal Ridge (AER) is a thickening of the epidermis
that forms along the outer rim of the limb bud
(of bird, mammal, reptile,
frog and fish embryos - but NOT on limb buds of salamanders!)
In frog embryos, the AER is comparatively small and indistinct, and for many years, everyone believed that frogs didn't have an AER. But more careful studies found them. The scientific meeting at which this evidence was first presented was in Wales in 1971, & I was in the audience!
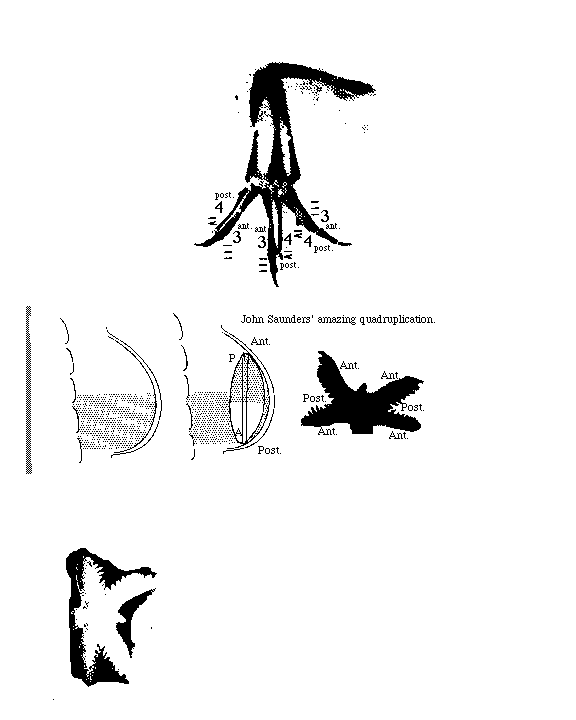
Surgical removal of the AER causes failure of the distal structures to develop. Cut the AER off early, then no elbow, forearm, wrist or hand. Cut the AER off later, then no wrist & hand.
This was discovered by Prof. John Saunders, who taught for many years at the State University of New York in Albany, and who visited this department a few years ago, and is one of the most stimulating scientists I have ever met. We had a prolonged barbeque lunch at Allen's out on route 86 north of town.

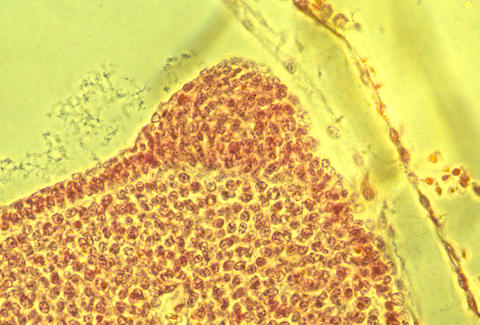

In sections of bird AERs, the epithelial cells seem to be pinched together at their basal ends, and the ridge is as much a "pucker" as a thickening.
Sections through mouse embryo AERs show a different geometry. It is definitely a thickening, but with some indication of contraction at Both basal and apical surfaces.
Here is a photograph of an oblique, or slanted section through a chicken AER.
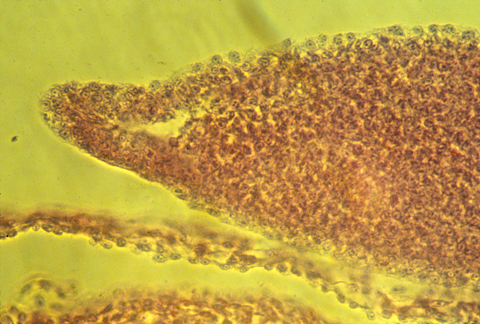
The developing tail fins of fish have a similar thickened ridge.
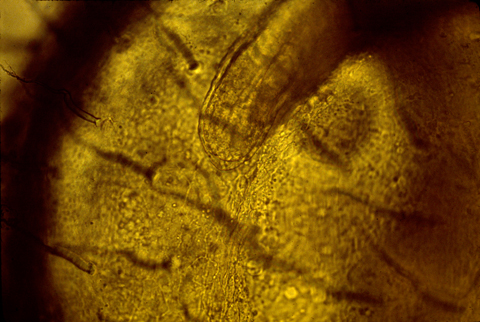
Grafting an extra AER can cause a double wing to form. A mutation in chickens causes double AERs and double wings!
But the AER doesn't itself become any these structures.
It somehow signals to them to form.
There has been much research and several theories on this subject.
Fibroblast growth factor 10 seems to be the key signal molecule.
FGF10 can replace the effect of the AER;
If you cut off the AER, & put a bead coated with FGF in its place
then the distal limb structures will form more or less normally.
(If anybody has done this with snake embryos, I haven't heard about it!
Probably, snakes still have the genes for legs, and maybe the signal receptors.)
Legless lizards and legless salamanders might also be subject to such experiments.
Another interesting fact is that salamanders (especially newts) are the only kinds of vertebrates that can regenerate cut off legs, and also are the only kind of vertebrate that don't form an AER.
However, during regeneration of salamander legs, an epithelial thickening is formed at the tip of the regenerating limb stump! This thickening is not elongated, however.
I suggest to you that some big conceptual breakthrough is waiting for you to discover!
If the AER cells are a special shape, does their shape mean they are more contractile, or what? Or is it an evolutionary remnant of fish fin shapes?
The proximo-distal axis is considered to be controlled by the AER.
The anterior-posterior axis can be reversed by grafting cells from the back side of the limb bud to the front side, which is called the "Zone of Polarizing Activity" (ZPA)
What is the chemical basis of this? Is it retinoic acid? Mirror-image branching legs can be produced by putting retinoic acid onto the anterior side of an early limb bud.
And if you put RA on the regenerating tail stump of a frog, then one or several legs will grow instead of a new tail!
And putting RA on the regenerating leg stump of a salamander will cause the entire proximo-distal sequence of structures to be regenerated, even if only the hand or wrist had been cut off!
The newer evidence is that the sonic hedgehog protein controls the anterior-posterior axis formation.
Organization of fingers somehow depends on certain hox genes: Specifically hoxD 9, 10, 11, 12, 13
The spaces between fingers are made by programmed cell death = "apoptosis" activation of cytoplasmic enzymes, that digest cells from the inside out.
The webbing of a duck's foot results from a lack of apoptosis.
And by the way, many cancer chemotherapy drugs actually work by stimulating apoptosis, rather than by direct harm to the cancer cells. The drugs were intended to kill fast growing cells, but don't really work that way!
The dorso-ventral axis of limb development: Can be reversed by removing the entire limb bud ectoderm and turning it upside down. The key protein is thought to be one of the Wnt proteins.
Some facts to get your interest:
I) Newts and many other kinds of salamanders can regenerate their legs.
No other vertebrates can do this, except partial regeneration of limbs in frog tadpoles before metamorphosis.
Humans can sometimes regenerate the last digit of fingers and toes, IF the wound is NOT stitched up (which is standard practice).
Regenerating newt legs "re-grow" all the bones (i.e. cartilages), muscles, tendons, blood vessels, connective tissue and skin.
In the stump of the cut limb, cartilage, muscle and other cell type become indistinguishable, "dedifferentiating" back to what looks like their early embryonic state, forming a mass of cells called a blastema, in which rapid cell growth and mitosis replaces the lost cells.
Skeletal muscle cells, which are syncytial, subdivide back to individual cells before resuming mitosis.
It would be interesting to know whether this subdivision of multi-nucleate muscle cells
has the same contractile-ring mechanism as the ordinary cytokinesis that follow mitosis.
The myoblasts then undergo many cycles of mitotic cell divisions, and fuse back together.
II) Grafting of cartilage and muscle labeled with radioactive labeled DNA, and also the use of genetic markers like nucleolus number, consistently show that each cell remains "loyal" to their original differentiated cell type.
In other words the dividing cells in the blastema that had been cartilage cells
re-differentiate only into cartilage,
& the descendants of muscle cells only become muscle.
Most experimenters had not expected this, which make the results even more persuasive.
It also contradicts expectations that regeneration should have the same basic mechanism as the original formation of limbs, except if the original development was by sorting out of cells whose differentiation had already been decided, which nobody considers possible.
Limb regeneration is not thought of as a special case of sorting out by differentiated cells, I guess because there is no deliberate dissociation into single or randomly-mixed cells.
Nevertheless, consider that cartilage cells from which the humerus had previously been constructed separate into what seem to be undifferentiated cells, and then re-differentiate as the radius, ulna digits, wrist bones, as well as replacements for the lost parts of the humerus.
Furthermore, consider that the many muscles of the regenerated leg are made of cells that had been parts of different muscles of the upper limb.
Likewise: skin cells, blood vessels, connective tissue & nerves.
All are replaced by descendants of the same kinds of the cells in the stump of the cut limb.
III) Salamanders are the only vertebrates whose limb buds do not form an apical ectodermal ridge.
However, during regeneration of salamander, the stumps develop an epithelial thickening which is well-formed, but not elongated in the A-P axis (unlike the AER)
I can't figure out how to interpret this combinations of odd facts, but it must mean something.
IV) If you cut off just the "hand" part of a newt leg, then just that part will regenerate.
If you cut the leg off at the elbow or knee, then just the missing part will regenerate.
If you cut off the whole leg, than all the parts of will regenerate.
BUT: If you cut off just the "hand" part, and then paint the cut surface with retinoic acid, then a whole new leg will regenerate, starting over at the shoulder, and forming an extra elbow, etc.
AND: Newts can regenerate their tails.
BUT if you cut off the tail, and paint the cut surface with retinoic acid, guess what regenerates instead!
The more of a newt leg you cut off, the faster tissues regenerate; So that the total time to completion of regeneration is about the same when you cut off the whole leg as when you cut off just the tip.
V) a) The spaces between fingers and toes are created by the programmed cell death.
So the web of a duck's foot is the primitive condition.
A chicken's foot has separate toes because programmed cell death has removed the tissues in between.
b) The bases of limbs are "sculpted" by regions of programmed cell death.
c) The neck is narrowed by large areas of programmed cell death.
d) The space between your gums and your cheek and limb tissue is created by programmed cell death.
e) The self-destruction of tadpole tails is a classic example of programmed cell death.
f) >98% of lymphocytes self-destruct by programmed cell death. (normally)
(in part, this serves to kill anti-self lymphocytes, whose binding sites fit some normal body protein)
g) Defense against viral infections is achieved by inducing programmed cell death of infected cells.
h) Graft rejection occurs by induction of programmed cell death,
by mechanisms that are meant to induce death of virally-infected cells.
(As if the grafted cells were mistaken for self-cells infected with viruses.)
i) Many viruses defend themselves by producing proteins that mimic the body's own inhibitors of programmed cell death. (specifically mimics of the bcl-2 protein, which will be discussed below.
j) Much of the death of heart cells during heart attacks, and of brain cells during strokes is claimed to be programmed cell death (setting off self-destruction enzymes), rather than simple destruction.
Therefore, chemicals that inhibit programmed cell death might save many lives.
k) Many anti-cancer drugs are now believed to act by inducing programmed cell death, somewhat selectively in cancer cells, but not so much in normal cells.
Conversely, a whole series of carbon chains attached to phosphates were designed to reduce osteoporosis by either strengthening bone or promoting more bone formation, but are now believed to stimulate programmed cell death in osteoclasts (but unfortunately in lots of other cell types, in addition to osteoclasts, with nasty side-effects)
l) One specific form of cancer is known to be caused (mostly) by too much synthesis of a particular protein named bcl-2, which localizes around mitochondria, and blocks release of reactive molecules (that release being part of how cells self-destruct in programmed cell death.)
B-Cell Lymphoma-2, because this was the second protein to be discovered by close studies of chromosome translocations in human victims of non-Hodgkins lymphoma. There are other proteins called bcl-1, and bcl-3 ; the latter is a cyclin.
Any gene that gets translocated to the chromosome site next to an antibody gene will get made in large amounts in B-lymphocytes, who are "trying" to make antibodies. Note that the same translocation in any other differentiated cell type might have no effect.
m) A large percentage of nematode embryonic cells are eliminated by programmed cell death. (Mutant worms in which this cell death doesn't occur are almost normal. It isn't at all clear what good it does the worm to have so many programmed cell deaths!
A nematode gene was found that is very similar to the human bcl-2 proteins, and replacement of this worm gene by the human bcl-2 gene restores normal development!
n) Another nematode gene, without which programmed cell death will not occur, was found to code for an inactive precursor of a protein-digesting enzyme, called a caspase. Part of the molecule blocks the active site, but can be digested off by a caspase from which its block has already been removed. Thus, activation of one caspase molecule will activate others, in a chain reaction, and they digest the cell from the inside out. Almost all cells of multicellular animals have millions of caspase enzymes dissolved in the cytoplasm.
o) Embryologists had been studying programmed cell death since ~1900
but the word "apoptosis" wasn't invented until 1980.
[There are disagreements about whether the second P is silent, or not:
Apo_Tosis or A Pop Tosis The latter is probably correct.
But be prepared for someone to correct you, no matter which way you pronounce it.]
Necrosis is what you call it when cells just die. (Especially if pieces of dead cell are left) Apoptosis is when they self-destruct.
p) Higher plants have a different kind of self-killing method, that they use to resist infections by induction of death in areas surrounding germs, so as to wall them off. Prof. Dangl is a world leader in research on this important phenomenon.
q) For many years, researchers guessed that there would be several different mechanisms of programmed cell death in animal cells, but apparently caspases are used for all the different examples, from making gaps between fingers to killing anti-self lymphocytes and virally-infected cells.
The greatest researcher on programmed cell death was Prof. Glucksman, of Cambridge University who was a friend of mine and a very wise man, and used to show me slides of programmed cell deaths.
--------------------------------------------
Questions
I) What are four embryonic organs whose medio-lateral, anterior-posterior and dorso-ventral axes become irreversibly decided during early development?II) Are all three axes decided simultaneously, or one at a time, or sometimes one at a time, but other times simultaneously?
III) Does the answer to the preceding question differ between organs?
IV) Which axis is the last to become irreversibly fixed, which second, and which third?
V) What are two mesodermal organs that start development as two separate organs (right and left), but then their tissues fuse?
VI) Driesch discovered that very early echinoderm embryos will do what if the first two or the first four cells are separated?
VII) What happens if you put two one cell stage embryos together when they are at their one cell stage?
VIII) Compare these phenomena discovered by Driesch to what happens in normal development of the mesodermal organs in question V.
IX) Why branch into three when a limb bud is grafted backwards?
X) How could you produce a 6 legged salamander (or chicken)? (Perhaps as extras in a John Carter of Mars movie?)
XI) Draw a sketch of the Apical Ectodermal Ridge in a bird embryo. Contrast the AER structure in embryos of birds, mammals, frogs, salamanders, and fish.
XII) From what you already know about the order in which axes become irreversibly fixed, are EphA gradients determined before EphB gradients, or the reverse. (hint: which controls the dorso-ventral axis of retinal differences of adhesion?)
XIII) Based on what you know about pressures across flexible surfaces, and their expected equilibrium between Pressures, Tensions and Curvatures:
dP= Curvature x Tension + curvature x tension
Please estimate changes of tensions in different parts of a limb buds as it develops from a small hemisphere, into a complex limb bud.
XIV) Can you deduce what might be special about the tension in the surface membrane at the AER, relative to other parts of the skin? (There is more than one possibility.)
Can you invent experiments capable of proving and/or disproving which possibility is true?
XV) After I draw and describe what Nardi and Stocum discovered happens when salamander limb buds are cut off at different proximo-distal levels, and then grafted to different proximo-distal levels of another limb bud (that is still attached to the body), try to explain what patterns of contractile forces cause the base of the graft to be pulled longitudinally along the host limb bud.
Why do you think equilibrium become balanced only when the base of the graft has been pulled to the homologous part of the host limb?
i.e. wrist next to wrist;
elbow next to elbow;
shoulder next to shoulder
XVI) When a salamander limb bud regenerates, the muscles near the cut surface "dedifferentiate" (become undistinguishable from cells that had been skeletal cells), and then grow and divide until the cell mass is nearly as big as the amount of tissue removed. Then muscle cells re-differentiate (only) into muscle cells, and skeletal cells redifferentiate (mostly) into skeletal cells.
What do you conclude about the mechanism of pattern formation?
Is it by rearrangement of cells according to cell type?
Or is it by re-differentiation of cells according to position?
XVII) Compare these alternatives to what H. V. Wilson hypothesized about the reformation of functional anatomy by dissociated sponge cells.
XVIII) How are Nardi and Stocum's observations related to the question of rearrangement versus refifferentiation?
XIX) Can you figure out any logical reason why the same category of protein (Fibroblast Growth Factor) induces third limbs and also causes the Medio-Lateral axis of limb buds? (I am not sure myself why this should be true.)
XX) Would you expect that the paracrine protein "Sonic Hedgehog" controls which part of the retina becomes connected to which part of the brain?
XXI) What about the paracrine protein Wnt? What axis does it control in developing limb buds? What properties of the neural retina does this axis correspond to?
XXII) Invent a method for using fibroblast growth factor to change the result of the Nardi and Stocum experiment.
XXIII) If you sliced a salamander limb bud like a long boloney, and cultured each of the slices onto a thin sheet of rubber (to detect amounts and directions of contractility), what differences would you expect, based on the result of the Nardi and Stocum experiment?
XXIV) Can you figure out whether and how the results of the Nardi and Stocum experiment can be explained by Steinberg's Differential Adhesion Hypothesis?
Would the embryonic limb need to become more adhesive near the outer end? Or more adhesive near the basal end?
XXV) Could the same Nardi and Stocum effect be produced by gradients of strength of contractile strength of a developing limb bud?