Lecture notes for Monday, February 22, 2016
Amoeboid Locomotion
(And also "Crawling Locomotion" of embryonic and adult tissue cells)On the exams, you may be asked to identify which kind of amoeba is shown in a photograph, a diagram, or a video (or is described)
-
[for example: What are four kinds of amoeboid cells in which cytoplasmic flow occurs?]
answer: Amoeba proteus; Shelled amoebae; Physarum; White blood cells = Leucocytes
[What kind of "amoeboid" cell is shown in the following photograph?
[What is special and unusual about the kind of organism shown in this video?
[What are some similarities (and differences between the notochord and Dictyostelium stalks?
[If you wanted to study the molecular process of (for example: * Cytoplasmic flow; *Plasma membrane formation; * Assembly of actin protein monomers into actin fibers; * Exertion of strong shearing forces tangentially through plasma membranes, *Phagocytosis, etc.),
and for each of these and other phenomena, which kind of amoeboid cell would you choose to study, because of what special advantages.
I) Amoeba proteus; & also Chaos carolinense
Cytoplasmic flow. ("Fountain Zone")
Sol <--> Gel interconversion
"Sol" more liquid-like behavior of cytoplasm; flows up the center of a gel-like tube of stiffer cytoplasm.
Rear contraction theory of Mast; Fountain-zone theory of Robert Allen.
This "gelling" of amoeba cytoplasm is caused by polymerization of cytoplasmic actin
Forward transport of marker particles attached to the outer surface of the plasma membrane;
And rearward transport of attached particles where pseudopodia are retracting.
II) Shelled amoebae. Difflugia and others.
Difflugia amoebae use pseudopodia to reach out & pull in tiny sand grains, which they build into a hollow ball, inside of which the amoeba lives. Some other genera secrete transparent hollow spheres.
When being protruded outward, each small pseudopod has a fountain-zone pattern of cytoplasmic flow and sol-gel conversion. When these pseudopodia touch anything, they adhere and are somehow induced to contract.
This cycle of Protrusion - Adhesion - Contraction pulls the cell forward and pulls particles rearward.
Many or most textbooks claim that tissue cells (especially tissue culture cells) crawl by such a cycle of
Protrusion - Adhesion - Contraction; but you will see in a video that they really don’t.
III) Rolling Amoebae:
Adhering marker particles are pulled forward across the top, and rearward on the bottom.
(The plasma membrane moves in the pattern of a tank tread).
The forward movement of marker particles is exactly twice as fast as the forward movement of the front edge of that cell. This is exactly the speed of the forward movement of a tank tread.
IV) Dictyostelium: (and other Cellular Slime Molds)
Crawling locomotion is the most like tissue cells of any of these kinds of amoeboid organisms.
Move either as individual cells, phagocytizing bacteria, and then as multicellular "slugs". This aggregation is induced to occur when cells "attract" each other by chemotaxis; the attractant substance (for D. discoideum) is cyclic AMP.
Differentiation of amoebae to form stalk, and spores. (in about the proportions of a coconut palm tree)
Fruiting bodies (Meaning tree-like combination of stalk plus spores] can be as big as several hundred thousand amoeboid cells, and as small as 14 cells. An extreme example of dilation symmetry, comparable to what Driesch discovered with larvae of starfish and sea urchins (Echinoderms) 16-fold One Thousand-fold.
V) Physarum Highly multinucleate syncytia
Gigantic in comparison to Amoeba proteus!
Millimeters up to several centimeters in length.
Found in rotten wood and soil, and not under water.
Extremely fast and dramatic cytoplasmic flow
Also alternating contraction-relaxation of whole amoebae.
VI) Labyrinthula
video
Mostly live in salt water. Cause a major disease of sea grasses & major ecological changes.
Secrete extracellular layers of plasma membrane. (called "slime-ways")
Inner-most cells are football-shaped, and are bounded by a stiffer plasma membrane, outside of which thet secrete more layers of (flexible) plasma membrane.
These outer layers of plasma membrane were long believed to be non-living mucous slime.
Despite the ability of the extracellular layers of plasma membrane to extend and crawl, by itself, hundreds of microns beyond where any of the football-shaped "cells" have moved, scientists resisted the idea of multiple layers of plasma membrane. Everybody assumed that either the "footballs" were nuclei and the "slime-ways"
were living cytoplasm, or alternatively that the "slime-ways" were extracellular mucus, and the "footballs" were ordinary cells. Eventually, electron microscopy and time-lapse films proved that the extracellular membranes and cytoplasm were alive.
There are more different kinds of amoeboid organisms, such as foraminifera, and others waiting to be discovered.
++++++++++++++++++++++++++++++++++++++++++++++++++++++++++
Examples of crawling locomotion by cells of multicellular animals:
(These are just a few of many examples)
Primary and secondary mesenchyme of sea urchin embryos;
Primitive streak and Hensen's node;
Neural crest cells; including pigment cells in the skin.
Nerve "growth cones" whose locomotion creates axons, controls their paths & "wires" the nervous system..
Videos:
leucocyte crawling among red blood cells
a single cell on a rubber substratum
video of chick primitive streak
Functions served by the crawling locomotion of tissue cells
Wound healing; Wound closure.
Invasiveness of cancer cells (including penetration back out of blood vessels in metastasis)
Extension of capillaries into tissues.
(Hypothetical) Use of cell traction to realign type I collagen to form tendons, ligaments and skeletal muscles
What exerts the force in the locomotion of tissue cells?
(which has mostly been studied in tissue culture)
Actin polymerizes (molecules of actin protein diffuse through the cytoplasm, and are somehow induced to polymerize ("gel?")) along some parts of the edges of each cell.
This polymerization pushes outward thin stiff flat sheets called "lamellipodia". (And sometimes rod-like filopodia and small, hemispherical "blebs" or lobopodia.)
Stiff, polymerized sheets of actin get pulled rearward toward the middle of each cell.
Anything that adheres to the outer surface of the plasma membrane gets pulled rearward.
(Including micron-sized particles of styrene, metal, or anything) "retrograde transport"
(Particles get pulled rearward on both the "top" and the "bottom" of crawling cells.)

Cells cultured either on gels or on sheets of rubber pull rearward and distort these materials (see the video of a single cell on a rubber substratum, linked above).
The force that moves the particles and distorts the gels and rubber is called "traction".
Traction results from molecular links through the membrane, connecting flowing actin to outside objects.
There is also evidence that at least some parts of the plasma membrane itself flow rearward.
For example, when crawling tissue culture cells are infected with myxoviruses, the newly synthesized virus membrane proteins are first detectable along the front-most edge of lamellipodia.
Statistical studies of retrograde particle transport indicate viscous linkage between actin and external objects (instead of an ordinary elastic linkage). Brownian motion is superimposed on steady actin flow.
Mesenchymal and epithelial cell locomotion is directionally inhibited by cell-cell contact.
This was proven and quantitated in tissue culture by by several statistical correlations between contact versus cell speed and lower-than-random overlap between adjacent cells.
The term "contact inhibition" was invented by Michael Abercrombie specifically to refer to inhibition of cell locomotion, and NOT inhibition of cell growth or division. (cf Wikipedia article on contact inhibition)
Some cancerous cells overlap more randomly and their locomotion is less inhibited by cell-cell contact.
Many suspect that reduced contact inhibition is the reason for the greater invasiveness of cancer cells.
More research is needed about effects of cell-cell adhesion on actin assembly.
Inhibition of actin assembly and retrograde flow may cause contact inhibition.
Use of cell traction to realign type I collagen to form tendons, ligaments and skeletal muscles
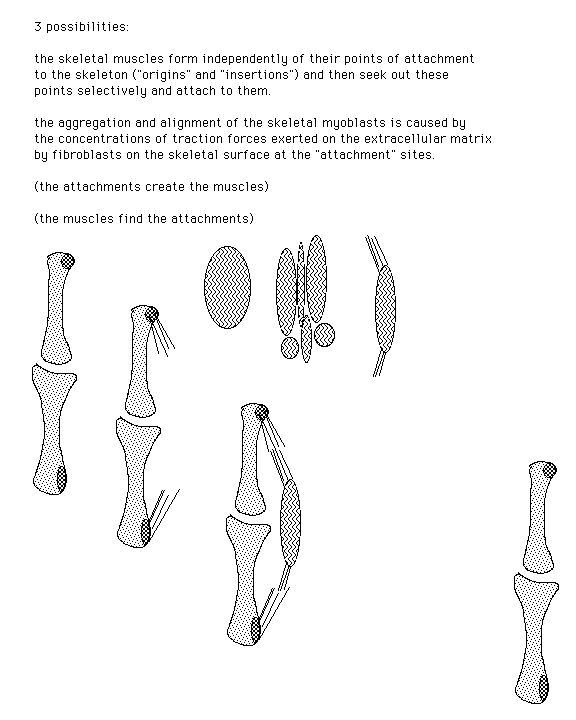
Diagram of muscle formation

This figure is from Stopak and Harris (1982). Connective Tissue Morphogenesis by Fibroblast Traction. Developmental Biology 90, 383-398. Leg bones from 10-day old chick embryos were put in a culture dish with pieces of thigh muscle in a collagen gel. The collagen becomes aligned between the muscle explants and the bones as a result of cell traction, forming an anatomical pattern that looks like a natural leg.
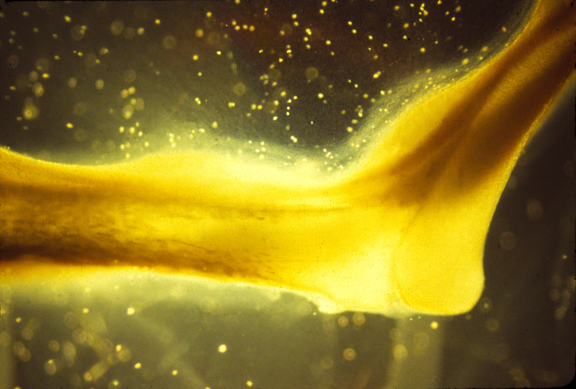
A color slide of an artificial muscle formed in this way.